ZIRGAN Dosing and Handling
ZIRGAN Dosing and Handling

Recommended dosing regimen1:
- One drop in the affected eye 5 times per day (approximately every 3 hours while awake) until the corneal ulcer heals
- Followed by 1 drop 3 times per day for 7 days
- Patients should be advised not to let the tip of the container touch any surface, as this may contaminate the ointment
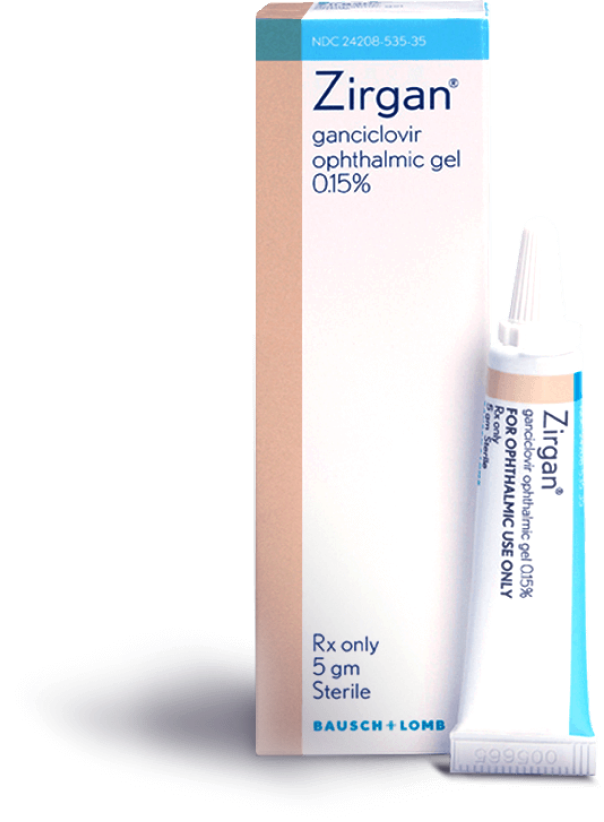
ZIRGAN is supplied as 5 grams of a topical ophthalmic gel containing 0.15% of ganciclovir in a polycoated aluminum tube.1
NDC number: 24208-535-35
There is no need for refrigeration (store between 59°F and 77°F). Do not freeze.1
If pain develops or if redness, itching, or inflammation becomes aggravated, the patient should be advised to consult their healthcare professional.
Help eligible patients save on ZIRGAN
Learn about a special savings offer from Bausch + Lomb.
See the detailsIndication
ZIRGAN® is an antiviral indicated for the treatment of acute herpetic keratitis (dendritic ulcers) in adults and pediatric patients aged 2 years and older.
Important Safety Information
- ZIRGAN® is indicated for topical ophthalmic use only.
- Patients should not wear contact lenses if they have signs or symptoms of herpetic keratitis or during the course of therapy with ZIRGAN®.
- Do not allow the tip of the container to touch any surface, as this may contaminate the ointment.
- If pain develops or if redness, itching, or inflammation becomes aggravated, the patient should be advised to consult a physician.
- Most common adverse reactions reported in patients were blurred vision (60%), eye irritation (20%), punctate keratitis (5%), and conjunctival hyperemia (5%).
- Safety and efficacy in pediatric patients below the age of 2 years have not been established.
Please see full Prescribing Information for ZIRGAN® or Click here for full Prescribing Information for ZIRGAN®.
You are encouraged to report negative side effects of prescription drugs to the FDA. Visit www.fda.gov/medwatch or call 1-800-FDA-1088.1-800-FDA-1088.
Reference: 1. ZIRGAN [package insert]. Bridgewater, NJ: Bausch & Lomb Incorporated.
Indication
ZIRGAN® (ganciclovir ophthalmic gel) 0.15% is a topical ophthalmic antiviral that is indicated for the treatment of acute herpetic keratitis (dendritic ulcers).
Important Safety Information
- ZIRGAN® is indicated for topical ophthalmic use only.
- Patients should not wear contact lenses if they have signs or symptoms of herpetic keratitis or during the course of therapy with ZIRGAN®.
- Most common adverse reactions reported in patients were blurred vision (60%), eye irritation (20%), punctate keratitis (5%), and conjunctival hyperemia (5%).
- Safety and efficacy in pediatric patients below the age of 2 years have not been established.
You are encouraged to report negative side effects of prescription drugs to the FDA. Visit www.fda.gov/medwatch or call 1-800-FDA-1088.1-800-FDA-1088.
Click here for full Prescribing Information for ZIRGAN®.
ZIRGAN® is a trademark of Laboratoires Théa Corporation used under license.
THIS WEBSITE IS INTENDED FOR U.S.
HEALTHCARE PROFESSIONALS ONLY.
Please choose an option below:



